Brush DC Drivers
Automotive Half-Bridge MOSFET Driver with ASIL Compliance and Enhanced Gate Drive
A4928
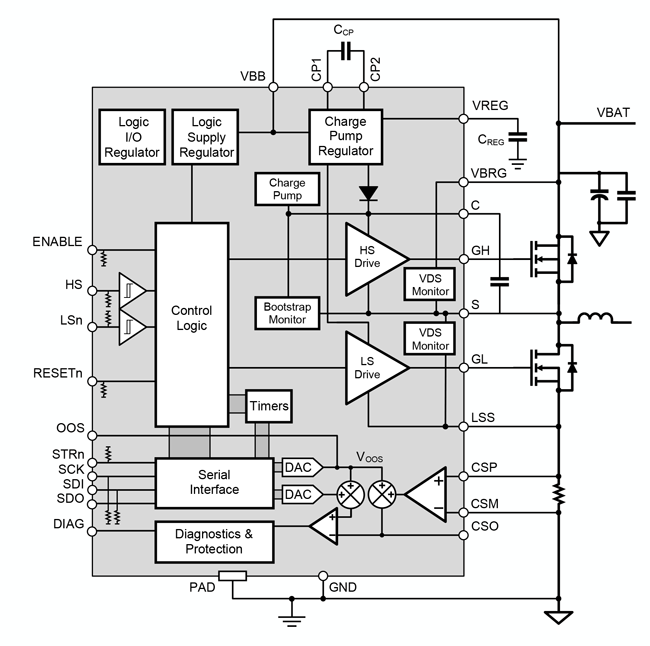
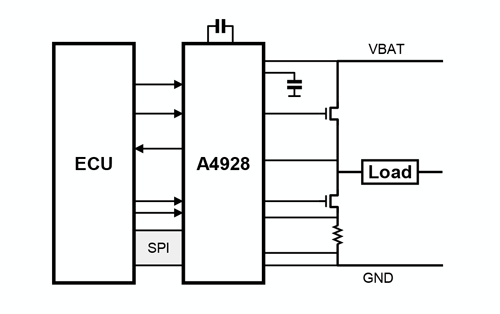
The A4928 provides robust and efficient control of high-power inductive loads in automotive applications, with ASIL compliance, enhanced gate drive, and integrated diagnostics.
Product Details
Product Details
Top Features
- Half-bridge MOSFET driver
- Bootstrap gate drive for N-channel MOSFET bridge
- Cross-conduction protection with adjustable dead time
- Charge pump regulator for low supply voltage operation
- 5.5 to 50 V supply voltage operating range
- 5 V CMOS Logic I/O
- SPI-compatible serial interface
- Bridge control by direct logic inputs or serial interface
- Programmable gate drive
- Current sense amplifier
- Programmable diagnostics
- Automotive AEC-Q100 qualified
- A2SIL™ product—device features for safety-critical systems