Brush DC Drivers
Digitally Programmable Dual Full-Bridge MOSFET Driver for Stepper and BLDC Motors
A3985
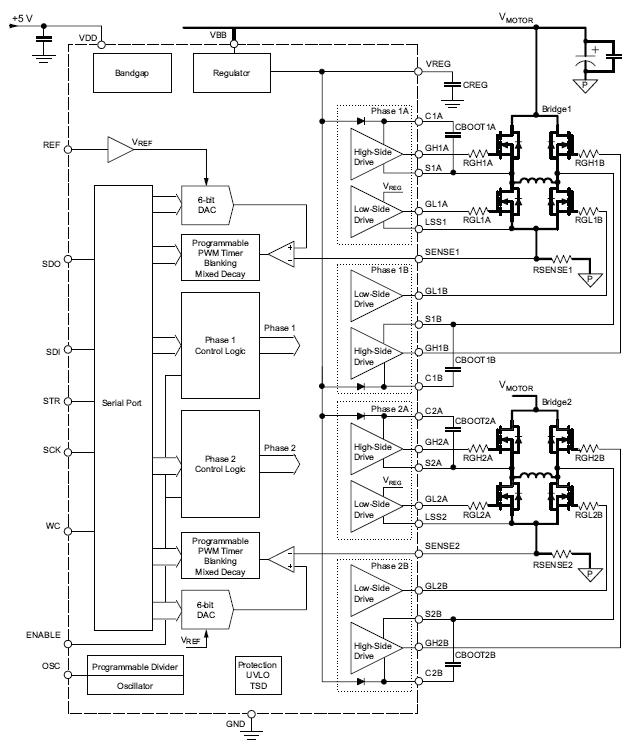
The A3985 offers a highly configurable solution for driving stepper and BLDC motors, featuring dual full-bridge gate drive, programmable PWM control, synchronous rectification, and integrated protection.
Product Details
Product Details
Top Features
- Serial interface for full digital control
- Dual full-bridge gate drive for N-channel MOSFETs
- Dual 6-bit DAC current reference
- Operation over 12 to 50 V supply voltage range
- Synchronous rectification
- Cross-conduction protection
- Adjustable mixed decay
- Fixed off-time PWM current control
- Low-current idle mode