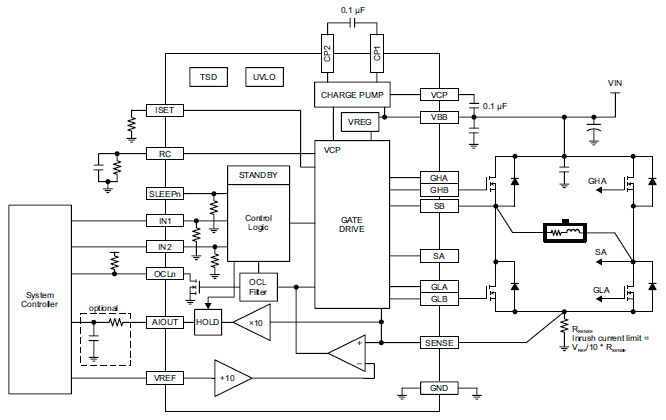
Serving as a replacement part for the A5957 Full-Bridge PWM Gate Driver. The A4955 provides a versatile and robust solution for driving all N-channel MOSFET bridges, featuring adjustable gate drive, synchronous rectification, and integrated protection.
Product Details
Product Details
Top Features
- Standard IN1/IN2 control logic
- Overcurrent indication
- Adjustable off-time and blank-time
- Adjustable current limit
- Adjustable gate drive
- Synchronous rectification
- Internal UVLO
- Crossover-current protection
- MOSFET VDS protection
- Voltage output proportional to load current
- A4955K is AEC-Q100 Grade 1 qualified
- Commercial temperature grade (A4955G: –40°C to 105°C)
- Automotive temperature grade (A4955K: –40°C to 125°C)