Brush DC Drivers
DMOS Stepper Motor Driver with Integrated Translator and Automatic Decay Selection
A3982
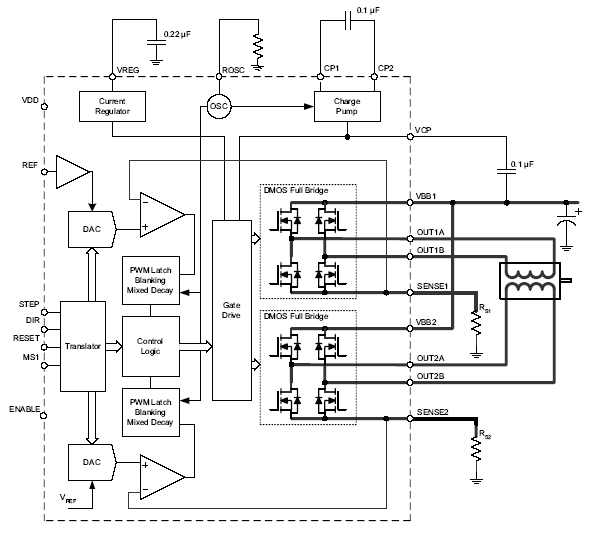
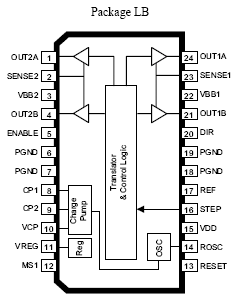
The A3982 simplifies stepper motor control with its integrated translator and automatic current decay mode selection, reducing design complexity and ensuring quiet, efficient operation.
Product Details
Product Details
Top Features
- Low RDS(ON) outputs
- Automatic current decay mode detection/selection
- Mixed and Slow current decay modes
- Synchronous rectification for low power dissipation
- Internal UVLO and thermal shutdown circuitry
- Crossover-current protection