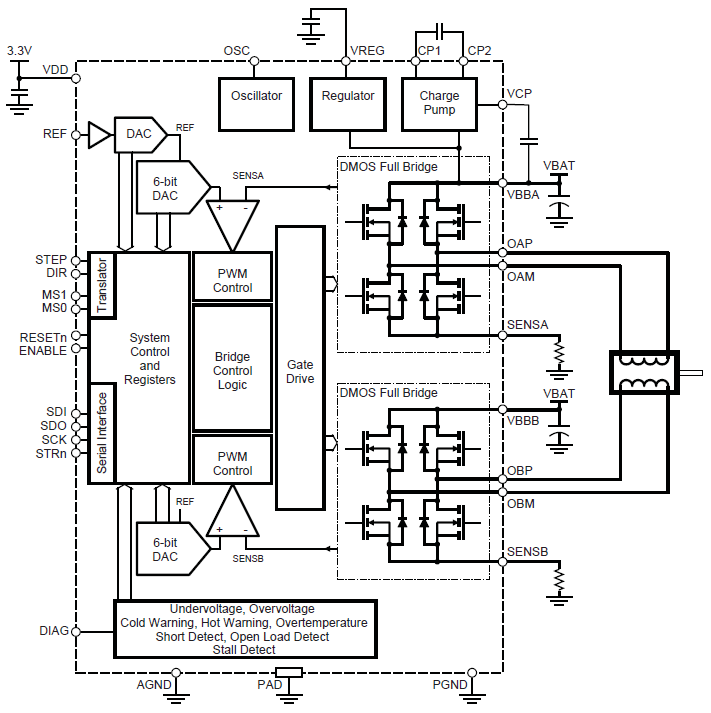
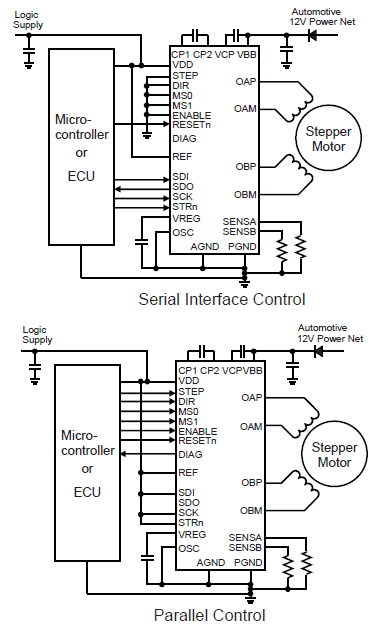
Serving as a replacement for the A3980, the A3981 provides highly configurable and reliable stepper motor control for automotive applications. Features SPI control, programmable current regulation, and diagnostic capabilities for enhanced system performance.
Product Details
Product Details
Top Features
- Peak motor current up to ±1.4 A, 28 V
- Low RDS(on) outputs, 0.5 Ω source and sink, typical
- Automatic current decay mode detection/selection
- Mixed, Fast, and Slow current decay modes
- Synchronous rectification for low power dissipation
- Internal OVLO, UVLO, and Thermal Shutdown circuitry
- Crossover-current protection
- Short circuit and open load diagnostics
- Hot and cold thermal warning
- Stall detect features
- SPI-compatible or simple Step and Direction motion control
- Highly configurable via SPI-compatible serial interface